AbstractBackground and PurposeWest syndrome is an epileptic encephalopathy of infancy. According to guidelines, adrenocorticotrophic hormone (ACTH) is probably effective for the short-term management of infantile spasm, but there is little uniformity in treatment due to variable response. This study has been done to evaluate the efficacy of pulse methylprednisolone as compared to ACTH in children with West syndrome.
MethodsChildren between 3 months to 24 months with the diagnosis of West syndrome were included and ACTH and pulse methyl prednisolone followed by oral prednisolone were given after randomization. Total duration of treatment was 6 weeks in both groups.
ResultsTotal 87 children were enrolled; 12 patients lost in follow up. Finally, 43 received ACTH and 32 received pulse methylprednisolone. In pulse methylprednisolone group, 28.13% showed 50–80% response, 28.13% showed 80–99% response and 21.87% patients showed 100% response. In ACTH group, 41.86% showed 50–80% response, 25.58% showed 80–99% response and only 3 (6.97%) patients showed 100% response. Methylprednisolone treatment regimen did not cause significant or persistent adverse effects.
ConclusionsPulse methylprednisolone followed by oral prednisolone for 6 weeks is as effective as ACTH. Thus, methylprednisolone therapy can be an important alternative to ACTH.
Key wordsWest syndrome, Methylprednisolone, Adrenocorticotropic hormonesIntroductionWest syndrome (WS) is a severe form of epilepsy which occurs in infancy.1 The infants present with a characteristic seizure manifested by myoclonic-tonic seizure (spasms) with a distinct form of electroencephalogram (EEG) pattern known as hypsarrhythmia. Developmental stagnation after the onset of spasm or delay is observed in almost all the infants. The spasms are of three types flexor, extensor or mixed.2 The prevalence is 0.249 cases/1,000 live births and the overall prevalence is 1 in every 10,000 children at the age of 10 years.3–5 Most children with WS manifest some degree of intellectual impairment and may develop other types of seizure.6–8
The preferred initial treatment for WS varies in different institute and geographic regions due to availability, cost and need of hospitalization.9–13 Till date, most of the publications suggested adrenocorticotropic hormones (ACTH) as first line treatment. However, there are disadvantages of administration of ACTH due to the high price, availability and adverse effects, particularly in a developing country setting. Moreover, ACTH did not cause complete remission of spasm in most of the previous studies, and thus it is not alone a persuasive option for treatment of WS.11–13 Other forms of corticosteroids are an alternative option; however, controversies are there which form of steroid should be given and how long.
Some recent studies suggested that intravenous pulse methyl-prednisolone (MP) followed by oral prednisolone is effective for seizure control in WS, which is safe.14,15 However, none of these studies were randomized controlled trial (RCT). On this ground, this study has been done to investigate the efficacy, safety and non-inferiority of pulse MP in WS in comparison to the standard drug ACTH. This is the first study in Bangladesh on WS where MP has been given in RCT.
MethodsThis study was done in the Department of Pediatric Neurology at a tertiary referral center in Bangladesh from July 2018 to June 2019 (1 year). Infants of 3 months to 24 months, who were diagnosed as WS on the basis of seizure semiology, developmental status and EEG, were taken as subjects. Informed written consent was taken from parents of all the patients. Children who had tuberous sclerosis complex with WS were excluded from the study as here the preferred treatment was vigabatrin. Patients who had underlying liver or kidney disorder, infection or electrolyte imbalance were also excluded from the study.
It was a randomized control trial. Initially children were evaluated through detail history and clinical examination. History related to seizure type, frequency, age at onset, perinatal details, family history, developmental history and detail ongoing treatment were noted. A base line complete blood count, electrolyte, liver function test, renal function test, blood glucose were done. In every case a 30 minutes sleep and awake EEG and neuroimaging (preferably magnetic resonance imaging of brain) was done. EEG reporting was done by an experienced pediatric neurophysiologist. Neuroimaging was reviewed by an expert neuro-radiologist. In suspected case, metabolic test (basic metabolic screening, tandem mass spectrometry [TMS], gas chromatography mass spectrometry [GCMS]) and genetic test (karyotyping, chromosomal microarray, clinical exome sequencing, whole exome sequencing) were done. Randomization was done by lottery method. Blinding was not possible as the administration of the two drugs were different.
Main outcome variable was proportion of subjects who had no seizure at the end of 6 weeks and side effects (clinical and biochemical). Children were reviewed in the inpatient department in first 7 days in both groups and then in outdoor on weekly basis for 5 weeks. At the end of the 6-week period, the average seizures per day in the preceding 2 weeks were compared in two groups. Parents were advised to contact in person or over phone if any emergency situation arrived.
Protocol of drugInjection methyl prednisolone was given 30 mg/kg/day in intravenous route in bolus and then oral prednisolone was given 2 mg/kg/day (6–30 days), 1mg/kg/day (31–36 days), and 0.5 mg/kg/day (37–42 days). Injection ACTH was given as follows 40 IU/day intramuscular in week 1–4, then 20 IU/day in week 5–6. Data was analyzed using statistical package for social science (SPSS) program version 22 for windows (IBM Corp., Armonk, NY, USA) and for all the analysis the p-value<0.05 was considered statistically significant.
ResultsDemographic and baseline characteristicsTotal 87 children were enrolled; 12 patients did not complete the treatment either due to failure to stay in the hospital or other comorbid conditions (infection, vomiting, etc.). At the time of inclusion, the mean age of MP group was 13.91±6.234 months and ACTH group was 11.63±6.321 months. The age of onset of spasm was 6.38±3.536 months and 5.14±3.005 months in MP and ACTH group, respectively. A male predominance was observed in both groups. Most of the patients in both groups had greater than 2 months lag to start the treatment. Majority of the patients showed delay in the development at the onset of the seizure. The most affected domain was motor, then cognition and speech (Table 1).
Etiology of WS in the studied subjectMost of the patients had structural etiology. In structural etiology, commonest cause was hypoxic ischemic encephalopathy (HIE) due to perinatal asphyxia. Other important etiologies were toxoplasmosis, other-syphilis, varicella-zoster, parvovirus B19, rubella, cytomegalovirus, and herpes infections (TORCH), neonatal hyperbilirubinemia, and malformation of brain (lissencephaly, schizencephaly, porencephaly, heterotopia, etc.). We also found some genetic etiology of WS (down syndrome, SLC1A1, CDKL5, SCN1A and SCN2A mutation). The metabolic etiologies were nonketotic hyperglycinemia, mitochondrial encephalopathy, methylmalonic acidemia and biotinidase deficiency. We were not able to identify the etiology of 4 cases. No statistically significant difference in etiology was observed between the groups (Table 2).
Birth history of the studied subjectMost of the subjects of both groups were term; only 6.25% of the MP group and 9.3% of ACTH group were preterm. About 9.3% of both groups were low birth weight and one patient had very low birth weight (Table 3).
EEG and neuroimaging profile of studied subjectIn this study, 53.125% of the MP group and 62.79% of ACTH group had hypsarrhythmia (classical and modified). Other types of EEG were multifocal discharge, diffuse encephalopathy, etc. Most common neuroimaging finding in both groups were cortical atrophy. Other findings were cystic encephalomalacia, calcification, basal Fatema K, et al. Methyl Prednisolone, Adrenocorticotropic Hormone, West Syndrome ganglia hyperintensity, infarction, neuronal migration defect, etc. (Table 4).
Response to drugIn MP group, 21.87% patients had complete remission of spasm. The rest of the patients of this group had partial remission. Twenty-eight percent of them had 80–99% cessation and another 28.13% had 50–80% cessation of spasm. The rest (21.87%) had <50% cessation of spasm. While in the ACTH treated group only three patients (6.97%) had complete cessation, 25.58% had 80–99% cessation, 41.86% had 50–80% cessation and the rest have <50% cessation of spasm. No statistically significant difference was found in both groups in this aspect (Fig. 1).
Adverse effects of drugsAdverse effects were evaluated during 6 weeks of drug treatment. None of the patient died during the treatment and no major adverse event occurred in both the groups. Patients of both groups have hypertension, irritability, infection, hyperglycemia, gastritis, etc. In ACTH group, the number of patients with hypertension and irritability was more than the MP group (Table 5).
Data processing and analysisData was statistically analyzed using SPSS program version 22 for windows and for all the analysis the p-value<0.05 was considered statistically significant. In addition, student t-test was done for normally distributed quantitative variables to measure mean and standard deviation.
DiscussionEarly diagnosis and early start of treatment is still the gold standard of optimal response of drugs in WS and the most used drugs for the treatment are ACTH, vigabatrin, corticosteroid and pyridoxine.16,17 However, ACTH has some potential side effects and may need prolonged hospital stay; moreover, for a developing country the drug is expensive.16,18 Whereas, MP followed by oral prednisolone is an important alternative protocol for treatment of WS and it is also cost effective. In this study, we found MP followed by oral prednisolone is as effective as ACTH. Moreover, it is safe to administer and there are minimal adverse effects.
In our study subject, 32 patients out of 75 received MP and 43 received ACTH. In both groups, around one fourth of the patients (21% in MP group and 25% in ACTH group) had <50% remission of seizure at 6 weeks. While 21.87% patients of MP got full remission of seizure, only 6.91% of the ACTH group had complete remission. Most of the patients of both groups had 50–99% remission. No statistically significant difference was found in seizure control in these groups. Our study result has similarity with that of Singhi et al.15 who found 50% remission in ACTH in 42.3% and in MP 22.2% at 6 weeks. However, their response rate at two weeks were around 50% in both groups (50% remission). Whereas, in their study Mytinger et al.14 reported that about 50% (5 out of 10) had rapid remission of spasm in 2–6 days with MP. Very limited RCT had been done on ACTH and MP. Regarding response of ACTH, it is variable in different study. In their study Newaz et al.19 reported that about 54% had complete remission with ACTH, while Fatema et al.9 in their RCT showed that about 51.61% of patients with WS had complete remission.
Early onset of treatment was an important factor for remission of seizure in WS in previous studies.6,20 Mytinger et al.14 observed 83% response to MP treatment in cases where treatment was started within 1 month of seizure onset.14 In our study there was a larger gap to start treatment. More than 80% study subjects got the treatment after a 2–month lag period. This may be a cause of decreased response to drugs.14,21,22
Here, we administered 2 mg/kg/day oral prednisolone after pulse MP to avoid the side effects of high dose steroid. In previous studies, it has been observed that MP followed by initial oral dose of prednisolone 4 mg/kg/day showed 83% response.14 Whereas, only oral prednisolone at the dose of 2 mg/kg/dose had remission rate of 8.3% to 28.6%. It is here to mention that Lux et al.13 used higher dose of oral steroid (5–8 mg/kg/day) and reported 70% remission rate. In spite of low dose of steroid, we got better response. This may be due to the initial pulse MP.23,24
Regarding the adverse effect of the drugs, there were more adverse effects in ACTH group. Here, more than half of the patients had irritability, 30.23% had hypertension and 27.90% had hyperglycemia. Other side effects were infection, hypernatremia and gastritis. Patient who got MP had less adverse effects and the most common was hypertension which was mostly resolved after the pulse was stopped. As high dose of oral corticosteroid suppresses the hypothalamic-pituitary-adrenal axis, there is possibility of severe and fatal infection, hypertension, hyperglycemia, etc.25,26 Thus, we gave low dose oral prednisolone after pulse MP. Moreover, most of MP treated children needed less hospital stay after the protocolled hospitalization of 7 days. Thus, costing of total treatment was less in MP group.
Regarding demography, a male predominance in the study subject was observed like other studies.27 Most of the patients had delay in development at the onset of seizure. Structural etiology was the most predominant risk factor; however, some genetic and metabolic cases were also found. We did not find any difference in etiology with related studies.9,10,27 Hypsarrhythmia was found in about half of the patient in both the groups like the other related studies in WS.28,29 Regarding the neuroimaging, the most common abnormality was cortical atrophy. Cystic encephalomalacia due to HIE, calcification, neuronal migration defect and infarction were also found in this study group. The findings match with the previous studies on neuroimaging of WS.30
This study demonstrates the comparable response rate of pulse MP with oral prednisolone and ACTH in WS in a developing country. The lack of severe side effect, affordability, ease of administration, less hospital stay is also favorable to administer MP in children with WS. However, further large-scale studies are needed to determine the effects.
Figure 1Response to drug in studied subject (n=75). MP, methylprednisolone; ACTH, adrenocorticotrophic hormone. 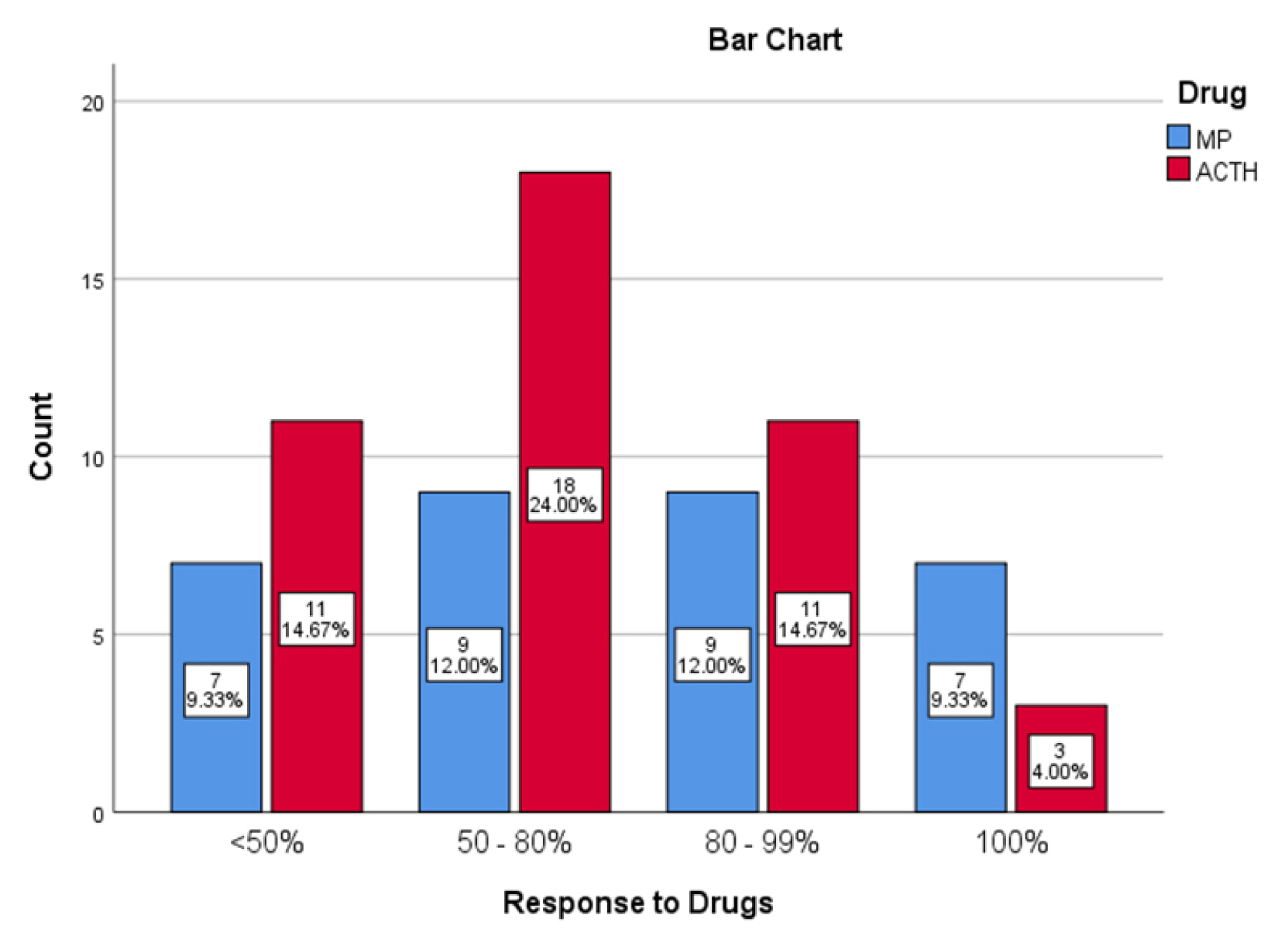
Table 1Demographic and baseline characteristics of the studied subject (n=75) Table 2Etiology of West syndrome in studied subject (n=75) Table 3Birth profile of studied subject (n=75) Table 4EEG and neuroimaging profile of studied subject (n=75) References2. Commission on Pediatric Epilepsy of the International League against Epilepsy. Workshop on infantile spasms. Epilepsia. 1992;33:195
3. Jia JL, Chen S, Sivarajah V, Stephens D, Cortez MA. Latitudinal differences on the global epidemiology of infantile spasms: systematic review and meta-analysis. Orphanet J Rare Dis. 2018;13:216
4. Lux AL. Latest American and European updates on infantile spasms. Curr Neurol Neurosci Rep. 2013;13:334
5. Riikonen R, Donner M. Incidence and aetiology of infantile spasms from 1960 to 1976: a population study in Finland. Dev Med Child Neurol. 1979;21:333–43.
7. Lombroso CT. A prospective study of infantile spasms: clinical and therapeutic correlations. Epilepsia. 1983;24:135–58.
8. Koo B, Hwang PA, Logan WJ. Infantile spasms: outcome and prognostic factors of cryptogenic and symptomatic groups. Neurology. 1993;43:2322–7.
9. Fatema K, Rahman MM, Akhter S, Shefa J. ACTH versus vigabatrin as first line treatment for West syndrome-a prospective study. Eur Acad Res. 2017;5:3760–70.
10. Rahman MM, Rahman SM, Fatema K. Clinical features and treatment outcome of West syndrome patients treated in a tertiary care hospital: Bangladesh perspective. Eur Acad Res. 2018;6:2638–51.
11. Lux AL, Edwards SW, Hancock E, et al. The United Kingdom Infantile Spasms Study (UKISS) comparing hormone treatment with vigabatrin on developmental and epilepsy outcomes to age 14 months: a multicentre randomised trial. Lancet Neurol. 2005;4:712–7.
12. Wanigasinghe J, Arambepola C, Sri Ranganathan S, Sumanasena S, Attanapola G. Randomized, single-blind, parallel clinical trial on efficacy of oral prednisolone versus intramuscular corticotropin on immediate and continued spasm control in West syndrome. Pediatr Neurol. 2015;53:193–9.
13. Lux AL, Edwards SW, Hancock E, et al. The United Kingdom Infantile Spasms Study comparing vigabatrin with prednisolone or tetracosactide at 14 days: a multicentre, randomised controlled trial. Lancet. 2004;364:1773–8.
14. Mytinger JR, Quigg M, Taft WC, Buck ML, Rust RS. Outcomes in treatment of infantile spasms with pulse methylprednisolone. J Child Neurol. 2010;25:948–53.
15. Singhi P, Gupta A, Rajpurohoit M, Saini AG, Soni V. Efficacy of pulse methylprednisolone vs adrenocorticotrophic hormone in children with West syndrome: an open-label pilot trial. Eur J Pediatr Neurol. 2017;21:e39
16. O’Callaghan FJ, Edwards SW, Alber FD, et al. Safety and effectiveness of hormonal treatment versus hormonal treatment with vigabatrin for infantile spasms (ICISS): a randomised, multicentre, open-label trial. Lancet Neurol. 2017;16:33–42.
17. Capal JK, Bernardino-Cuesta B, Horn PS, et al. Influence of seizures on early development in tuberous sclerosis complex. Epilepsy Behav. 2017;70:Pt A. 245–52.
19. Nawaz S, Gajre MP, Adhikari A, Shagufta . West syndrome: clinic etiological profile, and response to treatment in Western India. Int J Contemp Pediatr. 2020;7:1367–72.
20. Kivity S, Lerman P, Ariel R, Danziger Y, Mimouni M, Shinnar S. Long-term cognitive outcomes of a cohort of children with cryptogenic infantile spasms treated with high-dose adrenocorticotropic hormone. Epilepsia. 2004;45:255–62.
21. Glaze DG, Hrachovy RA, Frost JD, Kellaway P, Zion TE. Prospective study of outcome of infants with infantile spasms treated during controlled studies of ACTH and prednisone. J Pediatr. 1988;112:389–96.
22. Hrachovy RA, Frost JD, Kellaway P, Zion TE. Double-blind study of ACTH vs prednisone therapy in infantile spasms. J Pediatr. 1983;103:641–5.
23. Baram TZ, Mitchell WG, Tournay A, Snead OC, Hanson RA, Horton EJ. High-dose corticotropin (ACTH) versus prednisone for infantile spasms: a prospective, randomized, blinded study. Pediatrics. 1996;97:375–9.
24. Hancock E, Osborne J. Treatment of infantile spasms with high-dose oral prednisolone. Dev Med Child Neurol. 1998;40:500
25. Iyer A, Appleton R. Improving outcomes in infantile spasms: role of pharmacotherapy. Paediatr Drugs. 2016;18:357–66.
26. Coluccia D, Wolf OT, Kollias S, Roozendaal B, Forster A, de Quervain DJ. Glucocorticoid therapy-induced memory deficits: acute versus chronic effects. J Neurosci. 2008;28:3474–8.
27. Yeh HR, Kim MJ, Ko TS, Yum MS, You SJ. Short-term outcome of intravenous methylprednisolone pulse therapy in patients with infantile spasms. Pediatr Neurol. 2017;71:50–5.
28. Lagae L, Verhelst H, Ceulemans B, et al. Treatment and long term outcome in West syndrome: the clinical reality. A multicentre follow up study. Seizure. 2010;19:159–64.
|
|
||||||||||||||||||||||||||||||||||||