Introduction
The operational definition of status epilepticus is based on the length of time. Recent many trials were introduced, but 9–30% of status epilepticus remains to be refractory to treatment.1–3 Time-dependent pharmacoresistance is beneficial and therapeutic design can be used according to a defined protocol. For the stage of status epilepticus, impending, established, refractory status epilepticus can be used. Impending status is the initiation phase of status epilepticus, a disorder in which the duration of seizures is away from the norm of a single seizure. Established status is continuum of impending status, 30 min of continuous seizures. Refractory status is a term that is defined by the failure of adequate amounts of two intravenous drugs to stop seizures,4 and lasting more than 2 hours. Refractory status epilepticus develops in 23–44% of status epilepticus patients and has a mortality of 16–39%.2
In refractory status epilepticus, when phenytoin is failed to stop status epilepticus, barbiturate, benzodiazepines, ketamine, and propofol have been used according to treatment guideline of status epilepticus of ILAE and other recent reports.5–7 For continuous intravenous therapy for refractory status epilepticus, physicians chose especially midazolam and propofol, rather than a standard AED and then pentobarbital was chosen later in the therapy.7 Propofol is a ultrashort-acting sedative-hypnotic agent and has been considered as a promising drug, but may induce excitation and raise the risk of mortality.5 The propofol has medium frequency among the three drugs in an acute failure, breakthrough seizure, withdrawal seizure, hypotension and minimum risk of mortality.6 And propofol has disadvantages of frequent relapses when tapering, and risk of propofol infusion syndrome.8 We should monitor the side effects that are hypotension, bradycardia, rhabdomyolysis, and hypertriglycemia.5,9,10
The incidence of propofol infusion syndrome is about 39%, and mortality rate is up to 6%. Propofol infusion syndrome is characterized as the occurrence of acute bradycardia resistant to treatment and progressing to asystole, lipemia, hepatomegaly due to fat infiltration, severe metabolic acidosis, and muscle involvement with evidence of rhabdomyolysis or myoglobinuria and so on.11–13 According with other studies, the diagnosis of propofol infusion syndrome can be based on one or more those features11 or the presence of bradycardia and at least 1 sign of lipidemia, enlarged liver, metabolic acidosis, or rhabdomyolysis or myoblobulinuria.14,15 But those clinical features of propofol infusion syndrome are overlapped with those of refractory status epilepticus.16,17 If we don’t know of use of propofol, it is hard to distinguish propofol infusion syndrome from the unspecific clinical situations by refractory status epilepticus. Actually there are no studies that the presence of disease entity of propofol infusion syndrome in status epilepticus. The purpose of this study is to know whether the clinical findings of propofol infusion syndrome are due to use of propofol or refractory status epilepticus itself.
Subjects and Methods
Subjects
We enrolled all the patients who had admitted at Samsung Medical Center and diagnosed with status epilepticus from Jan. 2005 to Dec. 2009. Status epilepticus was defined as no recovery of mental status or seizures over 30 minutes, and RSE is defined as failure of first line and second line treatments with no time limit. Nonconvulsive status epilepticus was defined as presence of EEG of status epilepticus or improved patient with treatment of status epilepticus. We excluded patients without definite electrographic abnormalities or specific clinical features. And the patients who had not currently status epilepticus and under 16 years old were also excluded.
Thirty two patients were included. Female was 13 (40.6%) and male was 19 (59.4%). The median age was 28.5 years old (range, 16–81). Among them, 21 (65.5%) patients had refractory status epilepticus and 11 (34.4%) patients had status epilepticus that was terminated by the first- and second line treatments. The classification of status epilepticus was convulsive type in 31 patients and non-convulsive type in 1 patient. Among the 21 patients with refractory status epilepticus, 13 (61.9%) patients were treated with propofol and 8 (38.1%) patients with the other anesthetics such as midazolam and pentobarbital. The median days of hospitalization was 12.5 days (2–240 days). According to the outcome, 9 (28.1%) patients were death, 15 (46.9%) patients did not return to baseline state, and 11 (25.0%) patients returned to baseline state. The history of epilepsy was in 10 (31.3%) patients (Table 1).
Methods
We retrospectively reviewed the patients’ medical records. We collected the data on presence of previous history of epilepsy, clinical feature of status epilepticus (convulsive, non-convulsive), the causes of status epilepticus (infection, vascular, neoplasm, unknown), the numbers of concurrent antiepileptic drugs (AED) and clinical features of propofol infusion syndrome including metabolic acidosis, heart abnormalities, rhabdomyolysis, lipid change, hepatic change, renal change, death.
We evaluated the changes of clinical features on each category, based on the diagnostic criteria of Iyer et al.11 Metabolic acidosis was defined when one or more features of metabolic acidosis was found on arterial blood gases, heart changes when cardiac arrhythmia or asystole was on ECG, rhabdomyolysis when blood level of creatine kinase and myoglobulin were increased, lipid change when lipemia was detected on blood tests, hepatic change when elevated hepatic enzymes or abnormal ultrasonographic findings were found, renal change when urine output showed presence of oliguria, anuria, or blood test revealed increased creatinine or potassium. We categorized the outcomes of treatment into ‘return to baseline’, ‘do not return to baseline’ and ‘dead’.
We classified the patients with status epilepticus into established status epilepticus (ESE) and refractory status epilepticus (RSE) groups. And then the RSE group was further divided into propofol treatment (RSE-P) group and the other anesthetics treatment (RSE-O) group. The other intravenous anesthetics were midazolam and pentobarbital.
Analysis and statistics
We estimated the overall rate of the data of each clinical parameter. And then we compared those based first on the refractoriness of status epilepticus and then on use of propofol. So we analyzed the differences of clinical features between ESE and RSE groups. Also among RSE patients, RSE-P group was compared to RSE-O group. Finally we focused on the comparison of hospital days and treatment outcome between RSE-P and RSE-O groups. We used SPSS 14.0k for statistical analysis. We used Fisher’s exact two-tailed test for comparing sex, hypotension, metabolic acidosis, cardiac change, rhabdomyolysis, hepatic change, lipemia, renal change, death, previous history of epilepsy between groups. We also used Mann-Whitney U-test to analyze the age, numbers of AED. Analytical significance level was p<0.05.
Results
The propofol infusion therapy was performed in 13 patients. The method of propofol treatment was that the medial value of initial loading dose of propofol was 80 mg (range, 0–180 mg), the median value of maximal infusion rate of propofol was 8 mg/kg/hr (range, 0.3–15 mg/kg/hr), the median rate of maintenance of propofol infusion was 5.6 mg/kg/hr (range, 0.3–9.7 mg/kg/hr) and median duration of propofol infusion was 78 hours (range, 8–426 hours).
Analyzing the clinical features during the status epilepticus, hypotension was in 14 (56.3%) patients, metabolic acidosis in 7 (43.8%), cardiac change including arrhythmia in 15 (21.9%), rhabdomyolysis in 11 (46.9%), hepatic change in 6 (34.4%), lipid change in 8 (18.8%), and renal change in 10 (25.0%) (Fig. 1).
When we compared the clinical features between ESE and RSE, there was no significant difference in sex, age, metabolic acidosis, cardiac change, rhabdomyolysis, hepatic change, renal change, and number of antiepileptic drugs. The death rate was higher in RSE group (38.1%) than in ESE group (9.1%), while the previous epilepsy history was higher in ESE group (54.5%) than in RSE group (19.0%). But the death rate and previous epilepsy history did not show statistical significant differences (p≥0.05). Hypotension and dyslipidemia were much higher in RSE group (81.0%, 28.6%) than in ESE group (9.1%, 0.0%), which had statistical significant difference (p<0.001, p<0.05) (Table 2).
According to the anesthetic treatments, we compared the clinical features of refractory status epilepticus. When we compared RSE-P group to RSE-O group, there were not any significant differences in sex, age, hypotension, metabolic acidosis, cardiac change, rhabdomyolysis, hepatic change, lipid change, renal change, lipid change, the previous history of epilepsy and the number of antiepileptic drugs. In hypotension, RSE-P group (92.3%) had a higher rate than RSE-O group (62.5%). But it did not reach statistical significant difference (p>0.1) (Table 3).
We compared the duration of admission between the groups. When we compared SE group with RSE group, the hospital days were significantly longer in RSE group (median, range; 41, 2–240) than in ESE group (median, range; 7, 3–60) (p=0.012) (Fig. 2). However, there was no significant differences of hospital days between RSE-P group (median, range; 15, 2–229) and RSE-O group (median, range; 69, 9–240) (Fig. 3).
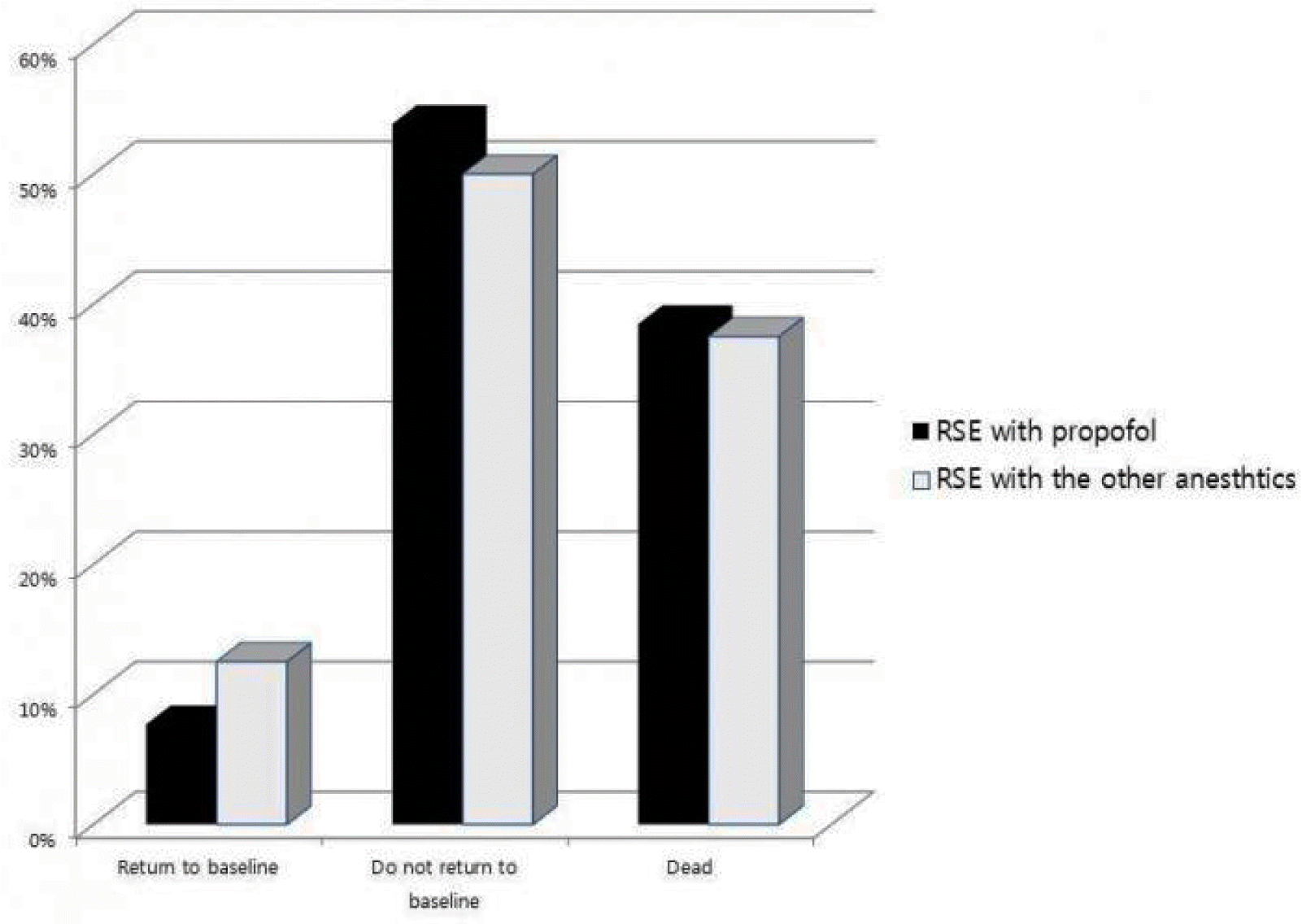
The outcomes after the treatment of status epilepticus showed statistically difference between ESE and RSE groups, having tendency to get worse outcome more in RSE group than in ESE group (p=0.007). However they did not between RSE-P and RSE-O groups. According to the classification of outcome after status epilepticus treatment, RSE group had 2 (9.5%) patients in “return to baseline”, 11 (52.4%) in “no return to baseline”, and 8 (38.1%) in “dead”, while ESE group had 6 (54.5%), 4 (36.4%), and 1 (9.1%) patients irrespectively (Fig. 4). However, when we compared RSE-P group with RSE-O group, the proportion of each category was not significant difference between them. The numbers of patients were 1 (7.7%), 7 (53.8%), and 13 (38.5%) in RSE-P group irrespectively, while they were 1 (12.5%), 4 (50.0%), and, 3 (37.5%) patients in RSE-O group irrespectively (Fig. 5).
Discussion
In our study, RSE showed longer hospital days and worse treatment outcome than ESE. That is not strange results because RSE is main cause of fatality in SE. Also RSE had significantly higher rate in hypotension and dyslipidemia than ESE. Interestingly our data suggest that cardiovascular problem and lipid profile changes could be prominent clinical parameters to indicating worse prognosis in RSE. However, Comparison of clinical features between RSE-P and RSE-O subgroups did not show any significant differences. This means that using propofol can cause propofol infusion syndrome, but RSE itself can cause the similar clinical features to propofol infusion syndrome.
Propofol has been used since the early 1980s for anesthesia in the operation room and sedation in the intensive care unit as well as currently for refractory status epilepticus.18 It is highly lipid soluble and metabolized in the liver, and has very short distribution and short elimination half-life (30–60 min) and little propensity to accumulate.19 The mechanism of action is postulated to be the modulation of GABA receptors at a site different from that targeted by benzodiazepines and barbiturates.20,21 High dose and prolonged treatment of propofol can cause propofol infusion syndrome. Some fatal cases have been reported.22,23 Typical clinical features of propofol infusion syndrome are lactic acidosis, arrhythmia, hypotension, cardiovascular failure, liver enlargement, increased liver enzymes, rhabdomyolysis, elevated serum creatine kinase, urea and potassium, hypertriglyceridemia, renal failure, oliguria, ketonuria, green or red-colored urine.24 Among them, lipemia, metabolic acidosis, and hepatic steatosis may have been caused by lipid infusion. The byproduct of liver conversion of fatty acids to ketone bodies could potentially contribute to metabolic acidosis. Metabolic acidosis could have also been caused by hypoperfusion secondary to propofol-induced bradycardia or precipitated by other concomitant medications (e.g., vasopressors). Muscle damage is indicated by the increase of serum creatine kinase and direct cardiac involvement is evidenced by the increase of serum cardiac troponin I.14 According to other study, propofol infusion syndrome was defined as suddenly developed and poorly controlled cardiac arrhythmia such as sudden bradycardia and asystole.13 Bradycardia or asystole is very important in diagnosis of propofol infusion syndrome, but it may not be unique results of using propofol. Those clinical features could be found in patients with RSE itself. So the abnormal chemistry profiles and cardiovascular changes might be considered not only in the findings of propofol treatment but also in the overall state of refractory status epilepticus.
Known risk factors of propofol infusion syndrome are using high dose propofol (>83 μg/kg/min), more than 48 hours, concomitant use of vasopressor.11,13 In this study, the mean infusion rate of propofol was 81.5 μg/kg/min. It was higher than the recommended dose of the ILAE guideline of treatment of SE (≥5 mg/kg/hr).5 The mean duration of treatment with propofol was 153 hour. It was longer than the period of duration as one of alleged risk factors of propofol infusion syndrome.9,23,25,26 Our use of propofol is more than enough to induce the propofol infusion syndrome. In other words, our cases are also having tendency to be very refractory to treatment.
In conclusion, our study did not demonstrate any significant differences in clinical features of propofol infusion syndrome between RSE-P and RSE-O groups. It may suggest that the clinical features of propofol infusion syndrome can be the clinical features of RSE regardless of using propofol. We need to be more careful in diagnosing propofol infusion syndrome whether those findings are due to propofol or RSE per se. This study is limited retrospective study and has the small numbers of patients, so that a more comprehensive prospective study could make the propofol infusion syndrome clear. Until further data become available, caution should be recommended when using high dose and long term propofol treatment.